1. Next Generation Thin Film Solar Cell Interface Engineering/Analysis
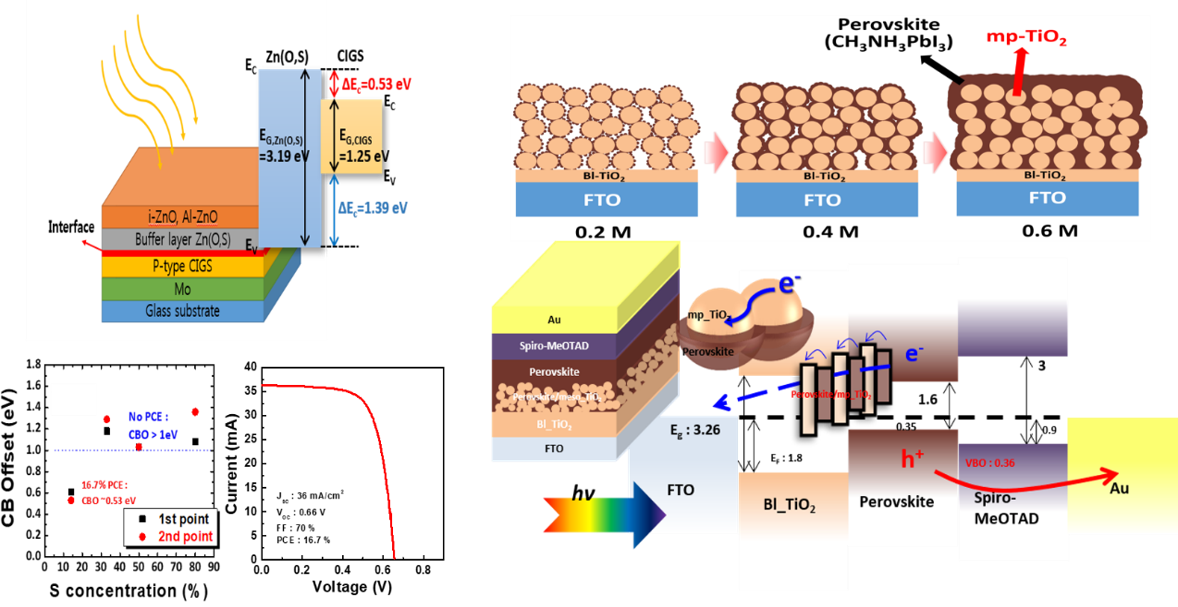
Next generation thin film solar cells such as CIGS and perovskite are of great interest for the researcher community to replace the conventional silicon based solar cells because of high process cost. However, stability and efficiency are main two parameters in order to commercialize the new generation solar cells. Interface engineering plays an important rule at the interface of material for the improvement in the efficiency. We are focusing on developing various deposition methods to reduce defects at the interface and performing optimization of solar cells through accurate spectroscopic (XPS, UPS, PL, SE) analysis.
- Further Readings
- Optimized electronic structure of a Cu(In,Ga)Se2 solar cell with atomic layer deposited Zn(O,S) buffer layer for high power conversion efficiency Modified Band Alignment Effect in ZnO-Cu2O Heterojunction Solar Cells via Cs2O Buffer Insertion”. J. Phys. D: Appl. Phys., 2018, 51, 055101 Depth Resolved Band Alignments of Perovskite Solar Cells with significant interfacial effects”. J. Mater. Chem. A, 2017, 5, 2563 “Plasmon-enhanced ZnO NR-Au NPs-Cu2O structure solar cells; effects and limitations”. Korean. J. Chem. Eng., 34(12), 3200-3207 (2017)
2. Solar Photocatalysis for Recyclable Clean Energy Fuel
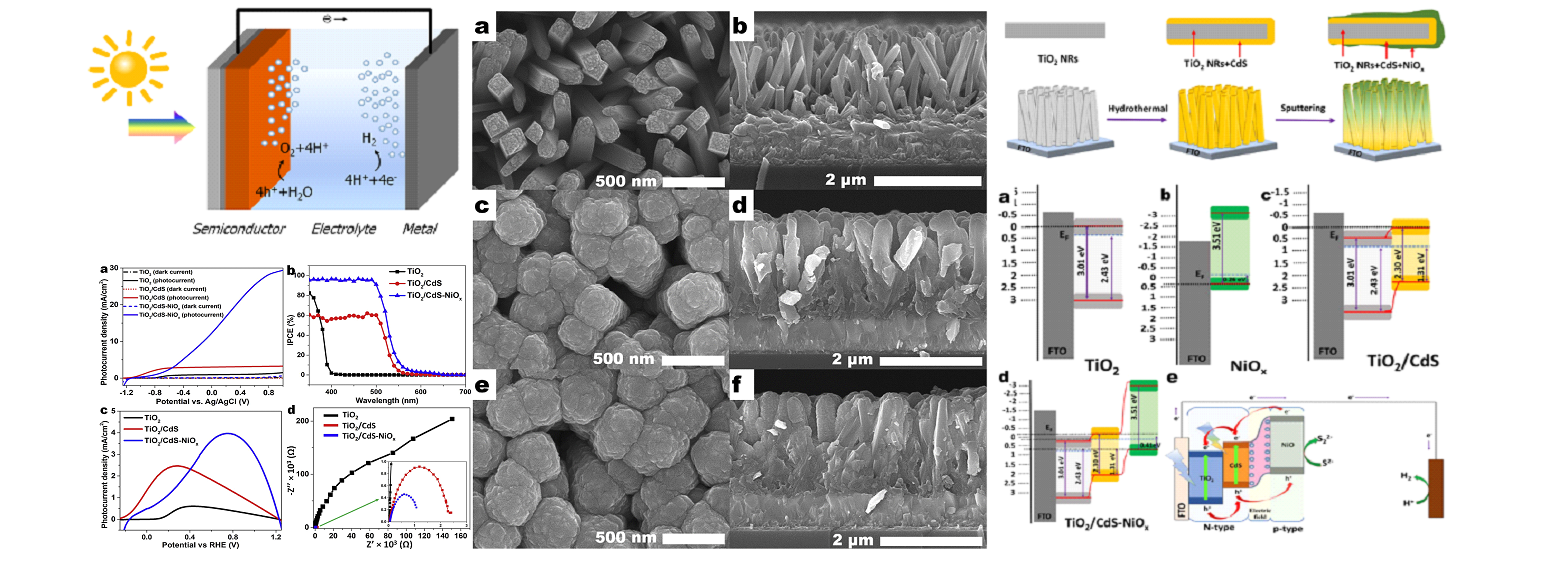
The water splitting is one of the most promising and cleaner method to produce hydrogen. In water splitting reaction, water and solar energy are used as source which are unlimited and environmentally cleaner. Our group synthesis various nanostructure metal oxide such as WOx, ZnO and SnSx for photocatalytic water splitting. To increase efficiency of hydrogen production reaction, various method that nanostructure, plasmon resonance effect and various additional layers are used.
- Further Readings
- “A nanoscale pn junction photoelectrode consisting of an NiOx layer on a TiO2/CdS nanorod core-shell structure for highly efficient solar water splitting”, Appl. Catal. B 250, 200-212 (2019) “Facile growth of compositionally tuned copper vanadate nanostructured thin films for efficient photoelectrochemical water splitting”, Appl. Catal. B 249, 235-245 (2019) “Enhancement of photoelectrochemical water splitting response of WO3 by Means of Bi doping”, J. Catal. 357, 127-137 (2018) “Insights into the electronic bands of WO3/BiVO4/TiO2, revealing high solar water splitting efficiency”, J. Mater. Chem. A 5 1455 (2017)
3. Methane Pyrolysis for CO2 Reduction and Hydrogen Production
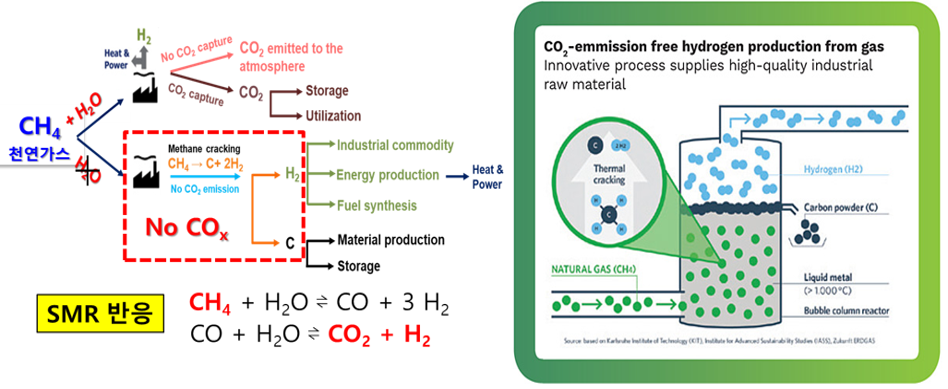
Currently, SMR process (steam methane reforming), is the main source of the hydrogen production however, production of CO2 gas as a byproduct is a big problem to the environment as global warming is the hot issue these days. To avoid this problem, methane pyrolysis method can be used for production of hydrogen without release of CO2. In methane pyrolysis method, molten metal catalyst directly decomposes methane to hydrogen and solid carbon, and unlike the solid catalyst, the molten metal catalyst is separated from carbon by density difference, thereby allowing a continuous process. Our goal is to develop catalyst materials to lower process temperature and to get high-quality solid carbon.
4. Flexible and Transparent Supercapacitors based on 2D Porous Materials
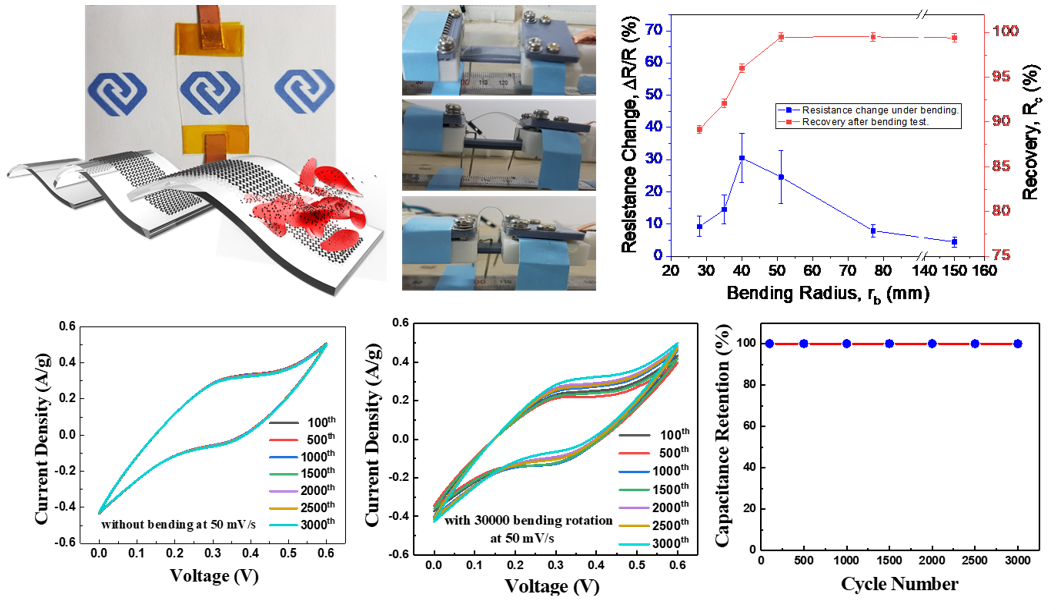
Wearable electronics have gained extensive attention. Having an important role in development of electronics, wearable power sources are of interest. Supercapacitors are of great interest for their applications in wearable electronics because of fast charge discharge time. 2D materials with outstanding large surface area, ultra-thin layer, high flexibility, and mechanically robustness meet most requirements for practical wearable supercapacitors. Specifically, carbon- based supercapacitors because of excellent mobility and ultrahigh surface area is one of the promising material for supercapacitors. For further, hybridization of carbon with other transition metal oxides is of interest for the improvement in electrochemical properties. Our group is focusing on the synthesis of new materials for wearable flexible energy storage device and multifunctional devices.